Hippocampal contributions to biologic, behavioral and cognitive deficits in autism: An update review
DOI:
https://doi.org/10.62063/rev-9Keywords:
autism spectrum disorder (ASD), behavior, biologic, cognitive, hippocampusAbstract
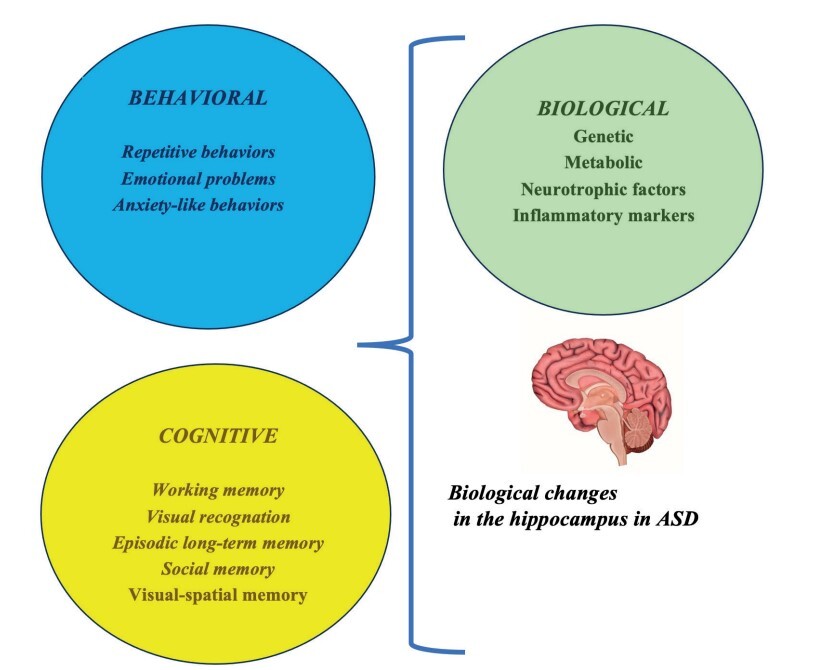
The most characteristic symptoms for the diagnosis of autism spectrum disorder (ASD) and the future life of the individual are deterioration in social communication and stereotyped or repetitive behaviors. ASD is associated with diverse atypical difficulties, including memory, learning, language, emotion, and cognitive impairment. Consequently, the hippocampus is important for memory, learning, language ability, emotional regulation, and cognitive mapping. Thus, the hippocampus plays an influential role in the pathophysiological mechanisms of ASD. Here, we provide an updated review of hippocampal structural and functional abnormalities and highlight the hippocampus as an important area for future research.
References
Ajram, L.A., Pereira, A.C., Durieux, A.M.S., Velthius, H.E., Petrinovic, M.M., & McAlonan, G.M. (2019). The contribution of [1H] magnetic resonance spectroscopy to the study of excitationinhibition in autism. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 89, 236-244. https://doi.org/10.1016/j.pnpbp.2018.09.010
Allsop, S.A., Wichmann, R., Mills, F., Burgos-Robles, A., Chang, C.J., Felix-Ortiz, A.C., Vienne, A., Beyeler, A., Izadmehr, E.M., Glober, G., Cum, M.I., Stergiadou, J., Anandalingam, K.K., Farris, K., Namburi, P., Leppla, C.A., Weddington, J.C., Nieh, E.H., Smith, A.C., Ba, D., Brown, E.N., Tye, K.M. (2018). Corticoamygdala Transfer of Socially Derived Information Gates Observational Learning. Cell, 173(6), 1329-1342.e18. https://doi.org/10.1016/j.cell.2018.04.004
Bangerter, A., Ness, S., Aman, M.G., Esbensen, A.J., Goodwin, M.S., Dawson, G., Hendren, R., Leventhal, B., Khan, A., Opler, M., Harris, A., & Pandina, G. (2017). Autism Behavior Inventory: A Novel Tool for Assessing the Core and Associated Symptoms of Autism Spectrum Disorder. Journal of Child and Adolescent Psychopharmacology, 27(9), 814-822. https://doi.org/10.1089/cap.2017.0018
Barbosa, A.G., Pratesi, R., Paz, G.S.C., Dos Santos, M.A.A.L., Uenishi, R.H., Nakano, E.Y., Gandolfi, L., & Pratesi, C.B. (2020). Assessment of serum BDNF levels as a diagnostic marker in children with autism spectrum disorder. Scientific Reports,10, 17348. https://doi.org/10.1038/s41598-020-74239-x
Barón-Mendoza, I., Mejía-Hernández, M., Hernández-Mercado, K., Guzmán-Condado, J., Zepeda, A., & González-Arenas, A. (2024). Altered hippocampal neurogenesis in a mouse model of autismrevealed by genetic polymorphisms and atypical development of newborn neurons. Scientific Reports, 14, 4608. https://doi.org/10.1038/s41598-024-53614-y
Bathina, S., & Das, U.N. (2015). Brain-derived neurotrophic factor and its clinical implications. Archives of Medical Science, 11(6), 1164-78. https://doi.org/10.5114/aoms.2015.56342
Bertoldi, M.L., Zalosnik, M.I., Fabio, M.C., Aja, S., Roth, G.A., Ronnett, G.V., & Degano, A.L. (2019). MeCP2 Deficiency Disrupts Kainate-Induced Presynaptic Plasticity in the mosaic fiber projections in the Hippocampus. Frontiers in Cellular Neuroscience, 13, 286. https://doi.org/10.3389/fncel.2019.00286
Barzegari, A., Amouzad Mahdirejei, H., Hanani, M., Esmaeili, M.H., & Salari, A.A. (2023). Adolescent swimming exercise following maternal valproic acid treatment improves cognition and reduces stress-related symptoms in offspring mice: Role of sex and brain cytokines. Physiology & Behavior, 269, 114264. https://doi.org/10.1016/j.physbeh.2023.114264
Camuso, S., La Rosa, P., Fiorenza, MT., & Canterini, S. (2022). Pleiotropic effects of BDNF on the cerebellum and hippocampus: Implications for neurodevelopmental disorders. Neurobiology of Disease, 163, 105606. https://doi.org/10.1016/j.nbd.2021.105606
Carlezon, W.A.Jr, Kim, W., Missig, G., Finger, B.C., Landino, S.M., Alexander, A.J., Mokler, E.L., Robbins, J.O., Li, Y., Bolshakov, V.Y., McDougle, C.J., & Kim, K.S. (2019). Maternal and early postnatal immune activation produce sex-specific effects on autism-like behaviors and neuroimmune function in mice. Scientific Reports, 9, 16928. https://doi.org/10.1038/s41598-019-53294-z
Chen, Y.S., Zhang, S.M., Yue, C.X., Xiang, P., Li, J.Q., Wei, Z., Xu, L., & Zeng, Y. (2022). Early environmental enrichment for autism spectrum disorder Fmr1 mice models has positive behavioral and molecular effects. Experimental Neurology, 352, 114033. https://doi.org/10.1016/j.expneurol.2022.114033
Coley, A.A., & Gao, W.J. (2018). PSD95: A synaptic protein implicated in schizophrenia or autism? Prog Neuropsychopharmacol Biological Psychiatry, 82, 187-194. https://doi.org/10.1016/j.pnpbp.2017.11.016
Cooper, R.A., & Simons, J.S. (2019). Exploring the neurocognitive basis of episodic recollection in autism. Psychonomic Bulletin & Review, 26, 163-181. https://doi.org/10.3758/s13423-018-1504-z
Cum, M., Santiago Pérez, J.A., Wangia, E., Lopez, N., Wright, E.S., Iwata, R.L., Li, A., Chambers, A.R., & Padilla-Coreano, N. (2024). A systematic review and meta-analysis of how social memory is studied. Scientific Reports, 14, 2221. https://doi.org/10.1038/s41598-024-52277-z
Desaunay, P., Guillery, B., Moussaoui, E., Eustache, F., Bowler, D.M., & Guénolé, F. (2023). Brain correlates of declarative memory atypicality in autism: a systematic review of functional neuroimaging findings. Molecular Autism, 14, 2. https://doi.org/10.1186/s13229-022-00525-2
Dionísio, A., Espírito, A., Pereira, A.C., Mouga, S., d’Almeida, O.C., Oliveira, G., & Castelo-Branco, M. (2024). Neurochemical differences in core regions of the autistic brain: a multivoxel 1H-MRS study in children. Scientific Reports,14, 2374. https://doi.org/10.1038/s41598-024-52279-x
Duarte-Campos, J.F., Vázquez-Moreno, C.N., Martínez-Marcial, M., Chavarría, A., Ramírez-Carreto, R.J., Velasco Velázquez, M.A., De La Fuente-Granada, M., & González-Arenas, A. (2024).Changes in neuroinflammatory markers and microglial density in the hippocampus and prefrontal cortex of the C58/J mouse model of autism. European Journal of Neuroscience, 59(1), 154-173. https://doi.org/10.1111/ejn.16204
Eissa, N., Azimullah, S, Jayaprakash P, Jayaraj RL, Reiner D, Ojha SK, Beiram R, Stark H, Łażewska D, Kieć-Kononowicz K, & Sadek B. (2019). The dual-active histamine H3 receptor antagonist and acetylcholine esterase inhibitor E100 ameliorates stereotyped repetitive behavior and neuroinflammation in sodium valproate-induced autism in mice. Chemico-Biological Interactions, 312, 108775. https://doi.org/10.1016/j.cbi.2019.108775
Elhamid, S.A.A., Alkherkhisy, M.M. & Kasem, R.E. (2024). Assessment of brain-derived neurotrophic factor levels in serum of children with autism spectrum disorders. Middle East Current Psychiatry, 31, 18. https://doi.org/10.1186/s43045-024-00403-y
Fetit, R., Hillary, R.F., Price, D.J., & Lawrie, S.M. (2021). The neuropathology of autism: A systematic review of post-mortem studies of autism and related disorders. Neuroscience & Biobehavioral Reviews, 129, 35-62. https://doi.org/10.1016/j.neubiorev.2021.07.014
Fuchs, C., Gennaccaro, L., Trazzi, S., Bastianini, S., Bettini, S., Lo Martire, V., Ren, E., Medici, G., Zoccoli, G., & Rimondini, R., Ciani, E. (2018). Heterozygous CDKL5 Knockout Female Mice Are a Valuable Animal Model for CDKL5 Disorder. Neural Plasticity, 9726950. https://doi.org/10.1155/2018/9726950
Fuentealba, C.R., Fiedler, J.L., Peralta, F.A., Avalos, A.M., Aguayo, F.I., Morgado-Gallardo, K.P., & Aliaga, E.E. (2019). Region-Specific Reduction of BDNF Protein and Transcripts in the Hippocampus of Juvenile Rats Prenatally Treated with Sodium Valproate. Frontiers in Molecular Neuroscience, 12, 261. https://doi.org/10.3389%2Ffnmol.2019.00261
Gage, F.H. (2019). Adult neurogenesis in mammals. Science, 364(6443), 827-828. https://doi.org/10.1126/science.aav6885
Georgiou, N., & Spanoudis, G. (2021). Developmental Language Disorder and Autism: Commonalities and Differences on Language. Brain Sciences, 11(5), 589. https://doi.org/10.3390/brainsci11050589
Girolamo, T., Shen, L., Monroe Gulick, A., Rice, M.L., & Eigsti, I.M. (2024). Studies assessing domains about structural language in autism vary in reporting practices and approaches to assessment: A systematic review. Autism, 28(7), 1602-1621. https://doi.org/10.1177/13623613231216155
Graham, S. F., Chevallier, O. P., Kumar, P., Trkolu, O. & Bahado-Singh, R. O. (2016). High-resolution metabolomic analysis of ASD human brain uncovers novel biomarkers of disease. Metabolomics, 12, 1-10.https://doi.org/10.1007/s11306-016-0986-9
Griffin, J.W., Bauer, R., & Scherf, K.S. (2021). A quantitative meta-analysis of face recognition deficits in autism: 40 years of research. Psychological Bulletin, 147(3), 268-292. https://doi.org/10.1037/bul0000310
Gifford, J.J., Deshpande, P., Mehta, P., Wagner, G.C., & Kusnecov, A.W. (2022). The Effect of Valproic Acid Exposure throughout Development on Microglia Number in the Prefrontal Cortex, Hippocampus and Cerebellum. Neuroscience, 481, 166-177. https://doi.org/10.1016/j.neuroscience.2021.11.012
Hanamsagar, R., Alter, M.D., Block, C.S., Sullivan, H., Bolton, J.L., & Bilbo, S.D. (2017). Generation of a microglial developmental index in mice and in humans reveals a sex difference in maturation and immune reactivity. Glia, 65(9), 1504-1520. https://doi.org/10.1002/glia.23176
Hao, S., Tang, B., Wu, Z., Ure, K., Sun, Y., Tao, H., Gao, Y., Patel, A.J., Curry, D.J., Samaco, R.C., Zoghbi, H.Y., & Tang, J. (2015). Forniceal deep brain stimulation rescues hippocampal memory in Rett syndrome mice. Nature, 526, 430-4. https://doi.org/10.1038/nature15694
Hashimoto, T., Yokota, S., Matsuzaki, Y., & Kawashima, R. (2021). Intrinsic hippocampal functional connectivity underlying rigid memory in children and adolescents with autism spectrum disorder: A case-control study. Autism, 25(7), 1901-1912. https://doi.org/10.1177/13623613211004058
Hitti, F.L., & Siegelbaum, S.A. (2014). The hippocampal CA2 region is essential for social memory. Nature, 508, 88-92. https://doi.org/10.1038/nature13028
Ilchibaeva, T., Tsybko, A., Lipnitskaya, M., Eremin, D., Milutinovich, K., Naumenko, V., & Popova, N. (2023). Brain-Derived Neurotrophic Factor (BDNF) in Mechanisms of Autistic-like Behavior in BTBR Mice: Crosstalk with the Dopaminergic Brain System. Biomedicines, 11(5), 1482. https://doi.org/10.3390/biomedicines11051482
Jasien, J.M., Daimon, C.M., Wang, R., Shapiro, B.K., Martin, B., & Maudsley, S. (2014). The effects of aging on the BTBR mouse model of autism spectrum disorder. Frontiers in Aging Neuroscience, 6, 225. https://doi.org/10.3389/fnagi.2014.00225
Jeon, S.J., Kwon, H., Bae, H.J., Gonzales, E.L., Kim, J., Chung, H.J., Kim, D.H., Ryu, J.H., & Shin, C.Y. (2022). Agmatine relieves behavioral impairments in Fragile X mice model. Neuropharmacology, 219, 109234. https://doi.org/10.1016/j.neuropharm.2022.109234
Kurochkin, I., Khrameeva, E., Tkachev, A., Stepanova, V., Vanyushkina, A., Stekolshchikova, E., Li, Q., Zubkov, D., Shichkova, P., Halene, T., Willmitzer, L., Giavalisco, P., Akbarian, S., & Khaitovich, P. (2019). Metabolome signature of autism in the human prefrontal cortex. Communications Biology, 2, 234. https://doi.org/10.1038/s42003-019-0485-4
Leisman, G., Melillo, R., & Melillo, T. (2023). Prefrontal functional connectivities in autism spectrum disorders: A connectopathic disorder affecting movement, interoception, and cognition. Brain Research Bulletin, 198, 65-76. https://doi.org/10.1016/j.brainresbull.2023.04.004
Li, G., Chen, M.H., Li, G., Wu, D., Lian, C., Sun, Q., Rushmore, R.J., % Wang, L. (2023). Volumetric Analysis of Amygdala and Hippocampal Subfields for Infants with Autism. Journal of Autism and Developmental Disorders, 53(6), 2475-2489. https://doi.org/10.1007/s10803-022-05535-w
Li, Y., Shen, M., Stockton, M.E., & Zhao, X. (2019). Hippocampal deficits in neurodevelopmental disorders. Neurobiology of Learning and Memory, 165, 106945. https://doi.org/10.1016/j.nlm.2018.10.001
Li, Z., Zhu, Y.X., Gu, LJ, & Cheng Y. (2021). Understanding autism spectrum disorders with animal models: applications, insights, and perspectives. Zoological Research, 42(6), 800-824. https://doi.org/10.24272/j.issn.2095-8137.2021.251
Libero, L.E., Reid, M.A., White, D.M., Salibi, N., Lahti, A.C., & Kana, R.K. (2016). Biochemistry of the cingulate cortex in autism: An MR spectroscopy study. Autism Research, 9(6), 643-57. https://doi.org/10.1002/aur.1562
Liu, S.H., Shi, X.J., Fan, F.C., & Cheng, Y. (2021). Peripheral blood neurotrophic factor levels in children with autism spectrum disorder: a meta-analysis. Scientific Reports, 11, 15. https://doi.org/10.1038/s41598-020-79080-w
Long, J., Li, H., Liu, Y., Liao, X., Tang, Z., Han, K., Chen, J., & Zhang, H. (2024). Insights into the structure and function of the hippocampus: implications for the pathophysiology and treatment of autism spectrum disorder. Frontiers in Psychiatry, 23,15, 1364858. https://doi.org/10.3389/fpsyt.2024.1364858
Meng, W. D., Sun, S. J., Yang, J., Chu, R. X., Tu, W., & Liu, Q. (2016). Elevated serum brainderived neurotrophic factor (BDNF) but not BDNF gene Val66Met polymorphism is associated with autism spectrum disorders. Molecular Neurobiology, 54, 1167-1172. https://doi.org/10.1007/s12035-016-9721-9
Pagni, B.A., Walsh, M.J.M., Ofori, E., Chen, K., Sullivan, G., Alvar, J., Monahan, L., Guerithault, N., Delaney, S., & Braden, B.B. (2022). Effects of age on the hippocampus and verbal memory in adults with autism spectrum disorder: Longitudinal versus cross-sectional findings. Autism Research, 10, 1810-1823. https://doi.org/10.1002/aur.2797
Raam, T., McAvoy, K.M., Besnard, A., Veenema, A.H., & Sahay, A. (2018). Author Correction: Hippocampal oxytocin receptors are necessary for discrimination of social stimuli. Nature Communications, 9(1), 552. https://doi.org/10.1038/s41467-018-02965-y
Rexrode, L.E., Hartley, J., Showmaker, K.C., Challagundla, L., Vandewege, M.W., Martin, B.E., Blair, E., Bollavarapu, R., Antonyraj, R.B., Hilton, K., Gardiner, A., Valeri, J., Gisabella, B., Garrett, M.R., Theoharides, T.C., & Pantazopoulos, H. (2024). Molecular profiling of the hippocampus of children with autism spectrum disorder. Molecular Psychiatry, 29, 1968–1979. https://doi.org/10.1038/s41380-024-02441-8
Sato, M., Nakai, N., Fujima, S., Choe, K.Y., & Takumi, T. (2023). Social circuits and their dysfunction in autism spectrum disorder. Molecular Psychiatry, 28, 3194–3206. https://doi.org/10.1038/s41380-023-02201-0
Segura, M., Pedreno, C., Obiols, J., Taurines, R., Pamias, M., Grunblatt, E., & Gella, A. (2015). Neurotrophin blood-based gene expression and social cognition analysis in patients with autism spectrum disorder. Neurogenetics, 16, 123–131. https://doi.org/10.1007/s10048-014-0434-9
Solmi, M., Song, M., Yon, D.K., Lee, S.W., Fombonne, E., Kim, M.S., Park, S., Lee, M.H., Hwang, J., Keller, R., Koyanagi, A., Jacob, L., Dragioti, E., Smith, L., Correll, C.U., Fusar-Poli, P., Croatto, G., Carvalho, A.F., Oh, J.W., Lee, S., Gosling, C.J., Cheon, K.A., Mavridis, D., Chu, C.S., Liang, C.S., Radua, J., Boyer, L., Fond, G., Shin, J.I., & Cortese, S. (2022). Incidence, prevalence, and global burden of autism spectrum disorder from 1990 to 2019 across 204 countries. Molecular Psychiatry, 27, 4172-4180. https://doi.org/10.1038/s41380-022-01630-7
Solomon, M., Ragland, J.D., Niendam, T.A., Lesh, T.A., Beck, J.S., Matter, J.C., Frank, M.J., & Carter, C.S. (2015). Atypical Learning in Autism Spectrum Disorders: A Functional Magnetic Resonance Imaging Study of Transitive Inference. Journal of the American Academy of Child & Adolescent Psychiatry, 54(11), 947-55. https://doi.org/10.1016/j.jaac.2015.08.010
Staudigl, T., Leszczynski, M., Jacobs, J., Sheth, S.A., Schroeder, C.E., Jensen O., & Doeller, C.F. (2018). Hexadirectional Modulation of High-Frequency Electrophysiological Activity in the Human Anterior Medial Temporal Lobe Maps Visual Space. Current Biology, 28(20), 3325-3329.e4. https://doi.org/10.1016/j.cub.2018.09.035
Sun, Y., Gao, Y., Tidei, J.J., Shen, M., Hoang, J.T., Wagner, D.F., & Zhao, X. (2019). Loss of MeCP2 in immature neurons leads to impaired network integration. Human Molecular Genetics, 28(2), 245-257. https://doi.org/10.1093/hmg/ddy338
Thomson, A.R., Pasanta, D., Arichi, T., & Puts, N.A. (2024). Neurometabolic differences in Autism as assessed with Magnetic Resonance Spectroscopy: A systematic review and metaanalysis. Neuroscience & Biobehavioral Reviews, 62, 105728. https://doi.org/10.1016/j.neubiorev.2024.105728
Trontel, H.G., Duffield, T.C., Bigler, E.D., Froehlich, A., Prigge, M.B., Nielsen, J.A., Cooperrider, J.R., Cariello, A.N., Travers, B.G., Anderson, J.S., Zielinski, B.A., Alexander, A., Lange, N., & Lainhart, J.E. (2013). Fusiform correlates of facial memory in autism. Behavioral sciences (Basel, Switzerland), 3(3), 348-71. https://doi.org/10.3390/bs3030348
Tsilioni, I., Patel, A.B., Pantazopoulos, H., Berretta, S., Conti, P., Leeman, S.E., & Theoharides, T.C. (2019). IL-37 is increased in the brains of children with autism spectrum disorder and inhibits human microglia stimulated by neurotensin. Proceedings of the National Academy of Sciences (PNAS), 116(43), 21659-21665. https://doi.org/10.1073/pnas.1906817116
Wan, L., Yang, G., & Yan, Z. (2024). Identification of a molecular network regulated by multiple ASD high-risk genes. Human Molecular Genetics, 33(13), 1176-1185. https://doi.org/10.1093/hmg/ddae058
Wang, Y., Zhang, Y.B., Liu, L.L., Cui, J.F., Wang, J., Shum, D.H, van Amelsvoort, T., & Chan, R.C. (2017). A Meta-Analysis of Working Memory Impairments in Autism Spectrum Disorders. Neuropsychology Review, 27, 46-61. https://doi.org/10.1007/s11065-016-9336-y
Zhao, H., Mao, X., Zhu, C., Zou, X., Peng, F., Yang, W., Li, B., Li, G., Ge, T., & Cui, R. (2022). GABAergic System Dysfunction in Autism Spectrum Disorders. Frontiers in Cell and Developmental Biology, 9, 781327. https://doi.org/10.3389/fcell.2021.781327
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Oznur Ozge Ozcan, Burcu Cevreli, Mesut Karahan, Muhsin Konuk

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.







